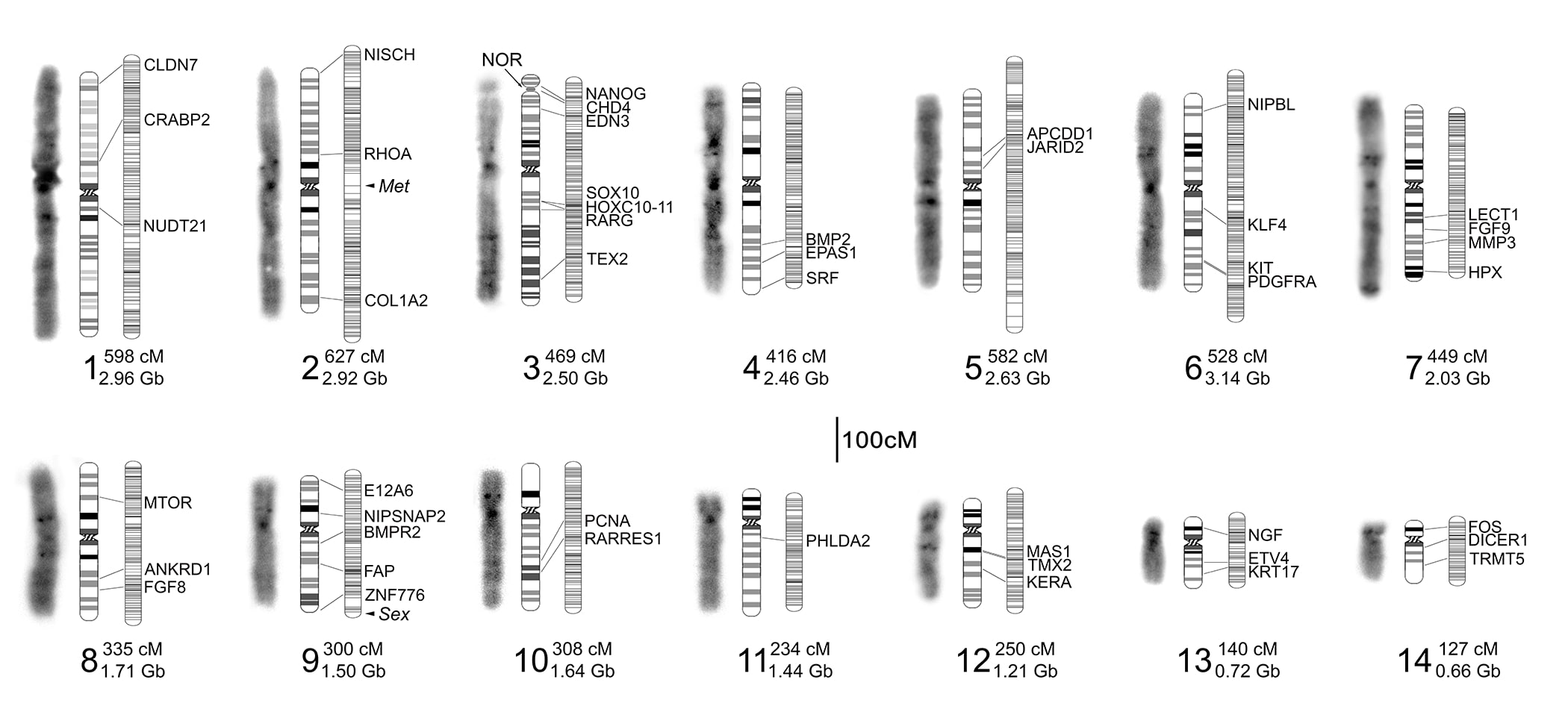
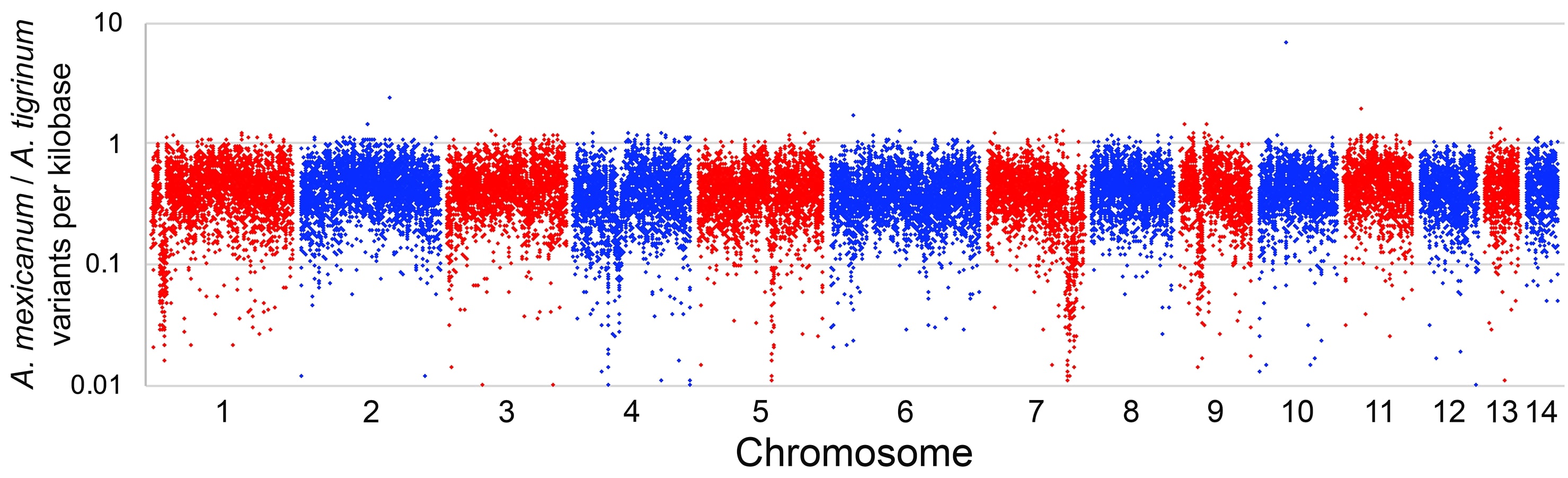
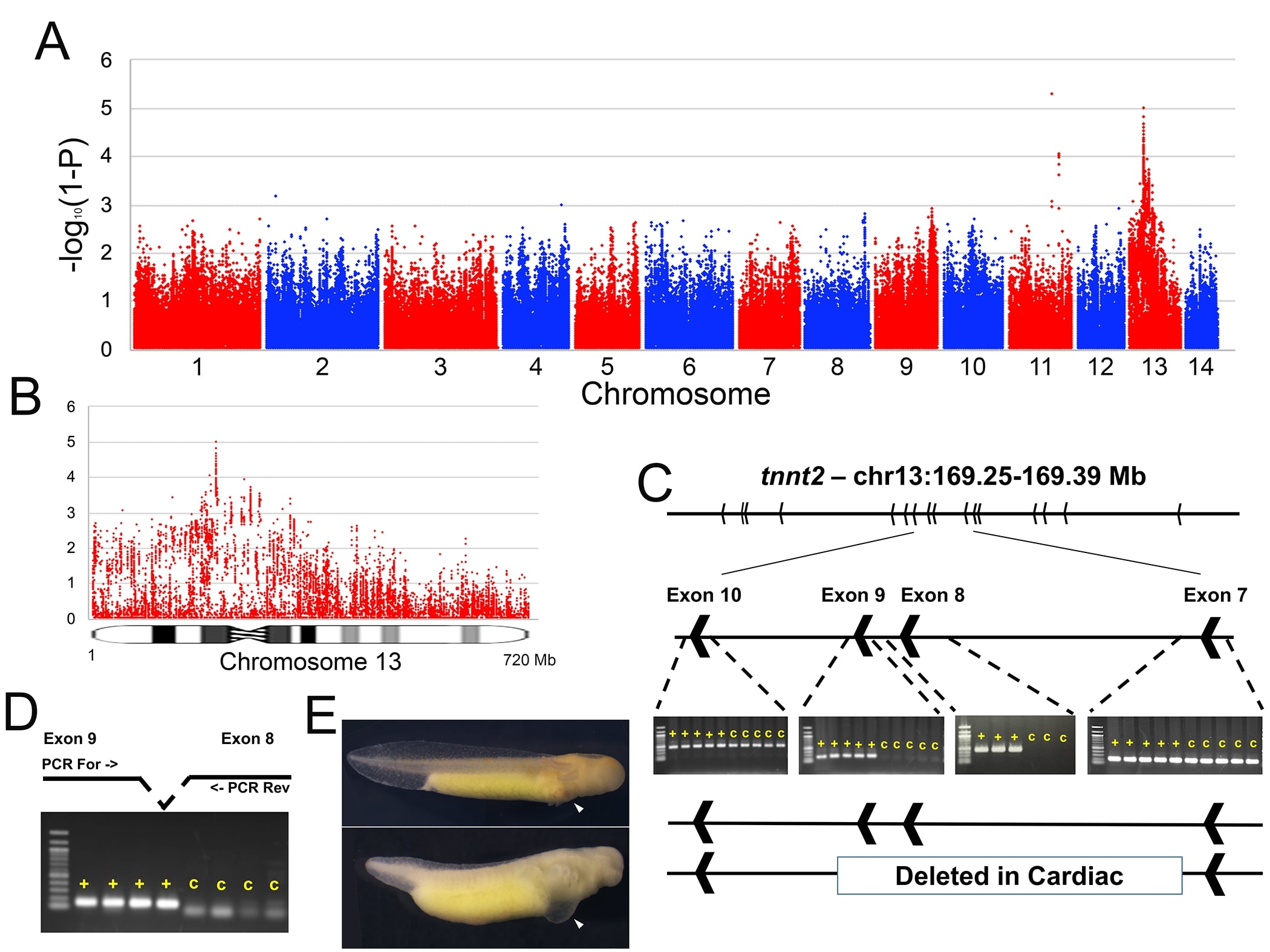
Mutant Gene List
The Genes
Albino (a)
The mutation for albinism was introduced into axolotls from an albino tiger salamander (Ambystoma tigrinum) female discovered in Minnesota (Humphrey, 1967; Hennen, 1977). Animals homozygous for this recessive gene completely lack the ability to synthesize melanin. Other pigments gre not affected, so an otherwise wild-type axolotl is yellow with reddish eyes and gills. Such an animal is sometimes called a "golden albino" (Frost, 1989). White-albino melanoid-albino, and axanthic-albino combinations are also possible.
The mutation for albinism was introduced into axolotls from an albino tiger salamander (Ambystoma tigrinum) female discovered in Minnesota (Humphrey, 1967; Hennen, 1977). Animals homozygous for this recessive gene completely lack the ability to synthesize melanin. Other pigments gre not affected, so an otherwise wild-type axolotl is yellow with reddish eyes and gills. Such an animal is sometimes called a "golden albino" (Frost, 1989). White-albino melanoid-albino, and axanthic-albino combinations are also possible.
Anemic (an)
This mutation is a non-lethal gene discovered in the Wistar white strain. Homozygotes develop temporary anemia when about 100 days old. Most recover and become normal adults. The onset of this anemia seems to be associated with the transition from larval to adult hemoglobin synthesis. In an/an animals adult hemoglobin synthesis is delayed. Frequently the anemia can be suppressed by treating the young axolotl with thyroxine (Tompkins, 1978) No carriers of this gene are identified in colony stocks today.
This mutation is a non-lethal gene discovered in the Wistar white strain. Homozygotes develop temporary anemia when about 100 days old. Most recover and become normal adults. The onset of this anemia seems to be associated with the transition from larval to adult hemoglobin synthesis. In an/an animals adult hemoglobin synthesis is delayed. Frequently the anemia can be suppressed by treating the young axolotl with thyroxine (Tompkins, 1978) No carriers of this gene are identified in colony stocks today.
Axanthic (ax)
The axanthic mutant is characterized by the absence of xanthophores and iridophores. Melanophores are normal, and otherwise wild-type animals are uniformly dark gray. Dark axanthic axolotls are not vigorous, and Frost reports a susceptibility to viral infections (Frost et al., 1986). Axanthic animals also homozygous for albino (a) are hardy animals. Older axanthic albinos are pale yellow, apparently due to riboflavins in the diet (Bukowski, et al., 1990).
The axanthic mutant is characterized by the absence of xanthophores and iridophores. Melanophores are normal, and otherwise wild-type animals are uniformly dark gray. Dark axanthic axolotls are not vigorous, and Frost reports a susceptibility to viral infections (Frost et al., 1986). Axanthic animals also homozygous for albino (a) are hardy animals. Older axanthic albinos are pale yellow, apparently due to riboflavins in the diet (Bukowski, et al., 1990).
Cardiac Non-function (c)
The cardiac mutation is a recessive gene primarily affecting the heart. The heart develops, but fails to contract. Subsequently, the embryos develop ascites. They do not feed and die soon after hatching. Homozygous cardiac individuals can be first identified about stage 36 by the swollen chest, poor gill development, characteristic red spot on the flank, and absence of a heartbeat. This gene has attracted much interest (e.g., Humphrey, 1972; Kulikowski and Manasek, 1978; Davis and Lemanski, 1987; Fransen and Lemanski, 1988; Lemanski and Fitzharris, 1989; Fransen and Lemanski, 1989; La France and Lemanski, 1994; Smith and Armstrong, 1990;1991; 1993a; 1993b) and considerable controversy (summarized by Armstrong in his 1985 review).
The cardiac mutation is a recessive gene primarily affecting the heart. The heart develops, but fails to contract. Subsequently, the embryos develop ascites. They do not feed and die soon after hatching. Homozygous cardiac individuals can be first identified about stage 36 by the swollen chest, poor gill development, characteristic red spot on the flank, and absence of a heartbeat. This gene has attracted much interest (e.g., Humphrey, 1972; Kulikowski and Manasek, 1978; Davis and Lemanski, 1987; Fransen and Lemanski, 1988; Lemanski and Fitzharris, 1989; Fransen and Lemanski, 1989; La France and Lemanski, 1994; Smith and Armstrong, 1990;1991; 1993a; 1993b) and considerable controversy (summarized by Armstrong in his 1985 review).
Eyeless (e)
Homozygous eyeless animals lack eyes (but may not completely lack the optic cup), have enhanced pigmentation, and are sterile. The gene is primarily in white Wistar stock (Humphrey, 1969) The gene has attracted much research interest (Van Deusen, 1973; Epp, 1978; Brun, 1978; 1990; 1993; Gruberg and Harris, 1981; Harris, 1979; 83; 84; Eagleson and Malacinski, 1986; Maccagnan and Muske, 1992).
Homozygous eyeless animals lack eyes (but may not completely lack the optic cup), have enhanced pigmentation, and are sterile. The gene is primarily in white Wistar stock (Humphrey, 1969) The gene has attracted much research interest (Van Deusen, 1973; Epp, 1978; Brun, 1978; 1990; 1993; Gruberg and Harris, 1981; Harris, 1979; 83; 84; Eagleson and Malacinski, 1986; Maccagnan and Muske, 1992).
Fluid Imbalance (f)
This mutation is characterized by an excess of fluid, first evident in tailbud stages, distending the head and later the trunk. It is semilethal. Surviving f/f females produce offspring which develop excess fluid precociously. All f/f offspring are non-viable. f is closely linked to gene g.
References: Humphrey, 1948; 1959; 1960; Dunson et al., 1971.
No carriers of this mutation are identified in the colony today.
This mutation is characterized by an excess of fluid, first evident in tailbud stages, distending the head and later the trunk. It is semilethal. Surviving f/f females produce offspring which develop excess fluid precociously. All f/f offspring are non-viable. f is closely linked to gene g.
References: Humphrey, 1948; 1959; 1960; Dunson et al., 1971.
No carriers of this mutation are identified in the colony today.
Gill Lethal (g)
This lethal recessive is closely linked to gene f (Humphrey, 1959). It is characterized by abnormal gill development and death within one week of hatching. The gills curve forward, are fragile, and have fewer filaments than do those of normal siblings. The mutant embryos are also darker and more yellow (Tompkins, 1970).
This lethal recessive is closely linked to gene f (Humphrey, 1959). It is characterized by abnormal gill development and death within one week of hatching. The gills curve forward, are fragile, and have fewer filaments than do those of normal siblings. The mutant embryos are also darker and more yellow (Tompkins, 1970).
Hand Lethal (h)
Larvae homozygous for this recessive mutant feed and grow until the forelimbs have three or four digits. At this time, the first digit has a thumb-like orientation. Some h/h larvae show a lateral curvature of the body. The larvae die very soon after reaching the 3-4 digit stage (Humphrey and Chung, 1977). No carriers of this mutation are identified in the colony today.
Larvae homozygous for this recessive mutant feed and grow until the forelimbs have three or four digits. At this time, the first digit has a thumb-like orientation. Some h/h larvae show a lateral curvature of the body. The larvae die very soon after reaching the 3-4 digit stage (Humphrey and Chung, 1977). No carriers of this mutation are identified in the colony today.
London Lethal (l)
Animals homozygous for this recessive gene display a disproportionately small head, small and poorly developed eyes, fragile gills with poorly developed secondary filaments, and undifferentiated limb buds just before hatching stage. They grow more slowly, and feed only a little after hatching. Some fluid accumulation in the head and trunk is common. They die a few weeks after hatching (Chung and Briggs, 1975). The gene was originally identified in white animals obtained from the colony of D.R. Newth by L.E. DeLanney.
Animals homozygous for this recessive gene display a disproportionately small head, small and poorly developed eyes, fragile gills with poorly developed secondary filaments, and undifferentiated limb buds just before hatching stage. They grow more slowly, and feed only a little after hatching. Some fluid accumulation in the head and trunk is common. They die a few weeks after hatching (Chung and Briggs, 1975). The gene was originally identified in white animals obtained from the colony of D.R. Newth by L.E. DeLanney.
Melanoid (m)
This recessive mutation is characterized by increased numbers of melanophores, reduced numbers of xanthophores, and the absence of iridophores. Adults are uniformly dark gray or black. The gene was discovered in descendants of wild stock imported from Mexico in 1961 (Humphrey and Bagnara, 1967) and has since been studied by several researchers (See, for example, Bagnara et al., 1978; Sawada and Dalton, 1979; Frost et al., 1984; Thorsteinsdottir and Frost, 1986; Frost et al., 1989.).
This recessive mutation is characterized by increased numbers of melanophores, reduced numbers of xanthophores, and the absence of iridophores. Adults are uniformly dark gray or black. The gene was discovered in descendants of wild stock imported from Mexico in 1961 (Humphrey and Bagnara, 1967) and has since been studied by several researchers (See, for example, Bagnara et al., 1978; Sawada and Dalton, 1979; Frost et al., 1984; Thorsteinsdottir and Frost, 1986; Frost et al., 1989.).
Microphthalmic (mi)
Homozygous microphthalmic larvae are identified around the time feeding begins. Their eyes are small, and the pigment cells often encroach upon the corneal surface. The effect may be asymmetric. They eat little if at all and soon die (Humphrey and Chung, 1977).
Homozygous microphthalmic larvae are identified around the time feeding begins. Their eyes are small, and the pigment cells often encroach upon the corneal surface. The effect may be asymmetric. They eat little if at all and soon die (Humphrey and Chung, 1977).
Pinhead (pi)
Pinhead is a craniofacial development mutant characterized at hatching by reduced interocular distances including instances of cyclopia. Primarily affected are the olfactory organs, eyes, forebrain, and oral cavity. The pharynx, otic vesicles, hindbrain, notochord, and gill are apparently normal. The gene appears to be a simple recessive, but penetrance is incomplete and variable. Rearing the embryos at 10 degrees C increases penetrance. Typically homozygotes exhibiting the phenotype are unable to eat and die soon after hatching. Mildly affected phenotypic pinheads may survive longer (Neff et al., 1987).
Pinhead is a craniofacial development mutant characterized at hatching by reduced interocular distances including instances of cyclopia. Primarily affected are the olfactory organs, eyes, forebrain, and oral cavity. The pharynx, otic vesicles, hindbrain, notochord, and gill are apparently normal. The gene appears to be a simple recessive, but penetrance is incomplete and variable. Rearing the embryos at 10 degrees C increases penetrance. Typically homozygotes exhibiting the phenotype are unable to eat and die soon after hatching. Mildly affected phenotypic pinheads may survive longer (Neff et al., 1987).
Lethal (r)
Larvae homozygous for this mutation become recognizable after they have been eating for a week or more. They have fragile gills with filaments of variable length, stop eating, become pale and slender, and soon die (Humphrey, 1964; Humphrey and Chung, 1978).
Larvae homozygous for this mutation become recognizable after they have been eating for a week or more. They have fragile gills with filaments of variable length, stop eating, become pale and slender, and soon die (Humphrey, 1964; Humphrey and Chung, 1978).
Short Toes (s)
Short toes is a recessive lethal mutation involving the kidneys, the Mullerian ducts and the limbs. It was first described by Humphrey (1967b) and has since been of interest especially to researchers who study limb development and regeneration (e.g., Mescher, 1993; Tsonis et al., 1993). Mutant animals can be readily identified by their short limbs and, especially, the reduced number of phalanges once front limb development is complete. Ascites, due to the incomplete development of the Mullerian ducts, becomes apparent only in the older juvenile. Homozygotes die of renal failure at about one year.
Short toes is a recessive lethal mutation involving the kidneys, the Mullerian ducts and the limbs. It was first described by Humphrey (1967b) and has since been of interest especially to researchers who study limb development and regeneration (e.g., Mescher, 1993; Tsonis et al., 1993). Mutant animals can be readily identified by their short limbs and, especially, the reduced number of phalanges once front limb development is complete. Ascites, due to the incomplete development of the Mullerian ducts, becomes apparent only in the older juvenile. Homozygotes die of renal failure at about one year.
Click on the images below for a larger picture.
 Homozygous adult showing characteristic ascites
Homozygous adult showing characteristic ascites
 Hand of homozygous short toes axolotl
Hand of homozygous short toes axolotl
Spastic (sp)
Hatchlings homozygous for this gene display sinusoid swimming and coiling patterns of movement characteristic of the embryonic stage. They fail to develop the "escape" or straight swimming patterns of normal larvae (Ide and Tompkins, 1975; Ide, 1978). No carriers of this mutation are identified in the colony today.
Hatchlings homozygous for this gene display sinusoid swimming and coiling patterns of movement characteristic of the embryonic stage. They fail to develop the "escape" or straight swimming patterns of normal larvae (Ide and Tompkins, 1975; Ide, 1978). No carriers of this mutation are identified in the colony today.
Stasis (st)
Mutants homozygous for this recessive gene are identified at hatching by their unusually red or pink livers, mild ascites, and pale color. Blood circulation ceases partially or completely. Most die within a week of hatching. A few survive longer (up to four months) (Humphrey and Chung, 1977).
Mutants homozygous for this recessive gene are identified at hatching by their unusually red or pink livers, mild ascites, and pale color. Blood circulation ceases partially or completely. Most die within a week of hatching. A few survive longer (up to four months) (Humphrey and Chung, 1977).
Twisted Gills (t)
Mutant larvae fail to feed and die, on average, seventeen days after hatching. Limb development is also retarded. Deterioration of the gills (responsible for the name) is apparently due to starvation (Armstrong et al., 1983).
Mutant larvae fail to feed and die, on average, seventeen days after hatching. Limb development is also retarded. Deterioration of the gills (responsible for the name) is apparently due to starvation (Armstrong et al., 1983).
Vasodilitation (v)
This mutation is a recessive semilethal gene characterized by vasodilitation at the time of hatching. Most die near to hatching. Survivors are smaller than their normal siblings. Eggs from v/v females arrest at blastula to neurula stages (Humphrey, 1962; Briggs and Humphrey, 1962). No carriers of this gene are identified in the colony today.
This mutation is a recessive semilethal gene characterized by vasodilitation at the time of hatching. Most die near to hatching. Survivors are smaller than their normal siblings. Eggs from v/v females arrest at blastula to neurula stages (Humphrey, 1962; Briggs and Humphrey, 1962). No carriers of this gene are identified in the colony today.
White (d)
The white mutation is a recessive gene affecting pigment cell distribution. A single white male among the first axolotls imported to Paris in 1868 became, apparently, the ancestor of all white axolotls in laboratories around the world (Newth, 1960). Homozygous individuals develop pigment cells for both black (melanophores) and yellow (xanthophores, iridophores) pigments, but they are restricted in distribution because they do not migrate normally off the neural crest during development. Many studies address this problem (for reviews see Armstrong, 1985; Frost, 1989. See also Lofberg et al., 1989a; 1989b; Thibaudeau et al., 1992; Epperlein and Lofberg, 1993.). White axolotls are white or pinkish in color, with black eyes. Frequently there is also dark pigment on the gills and, especially in older animals, dark speckling on the legs and tail.
The white mutation is a recessive gene affecting pigment cell distribution. A single white male among the first axolotls imported to Paris in 1868 became, apparently, the ancestor of all white axolotls in laboratories around the world (Newth, 1960). Homozygous individuals develop pigment cells for both black (melanophores) and yellow (xanthophores, iridophores) pigments, but they are restricted in distribution because they do not migrate normally off the neural crest during development. Many studies address this problem (for reviews see Armstrong, 1985; Frost, 1989. See also Lofberg et al., 1989a; 1989b; Thibaudeau et al., 1992; Epperlein and Lofberg, 1993.). White axolotls are white or pinkish in color, with black eyes. Frequently there is also dark pigment on the gills and, especially in older animals, dark speckling on the legs and tail.